Quick Read
- The Piezo1-STING axis acts as a bridge between physical mechanical stress and metabolic decay in diabetic skin cells.
- Cortistatin serves as an unrecognized inhibitory ligand that blocks this pathological signaling pathway.
- Targeting this regulatory axis offers a novel clinical strategy for managing chronic, non-healing diabetic wounds.
Uncovering the Piezo1-STING Signaling Axis in Diabetes
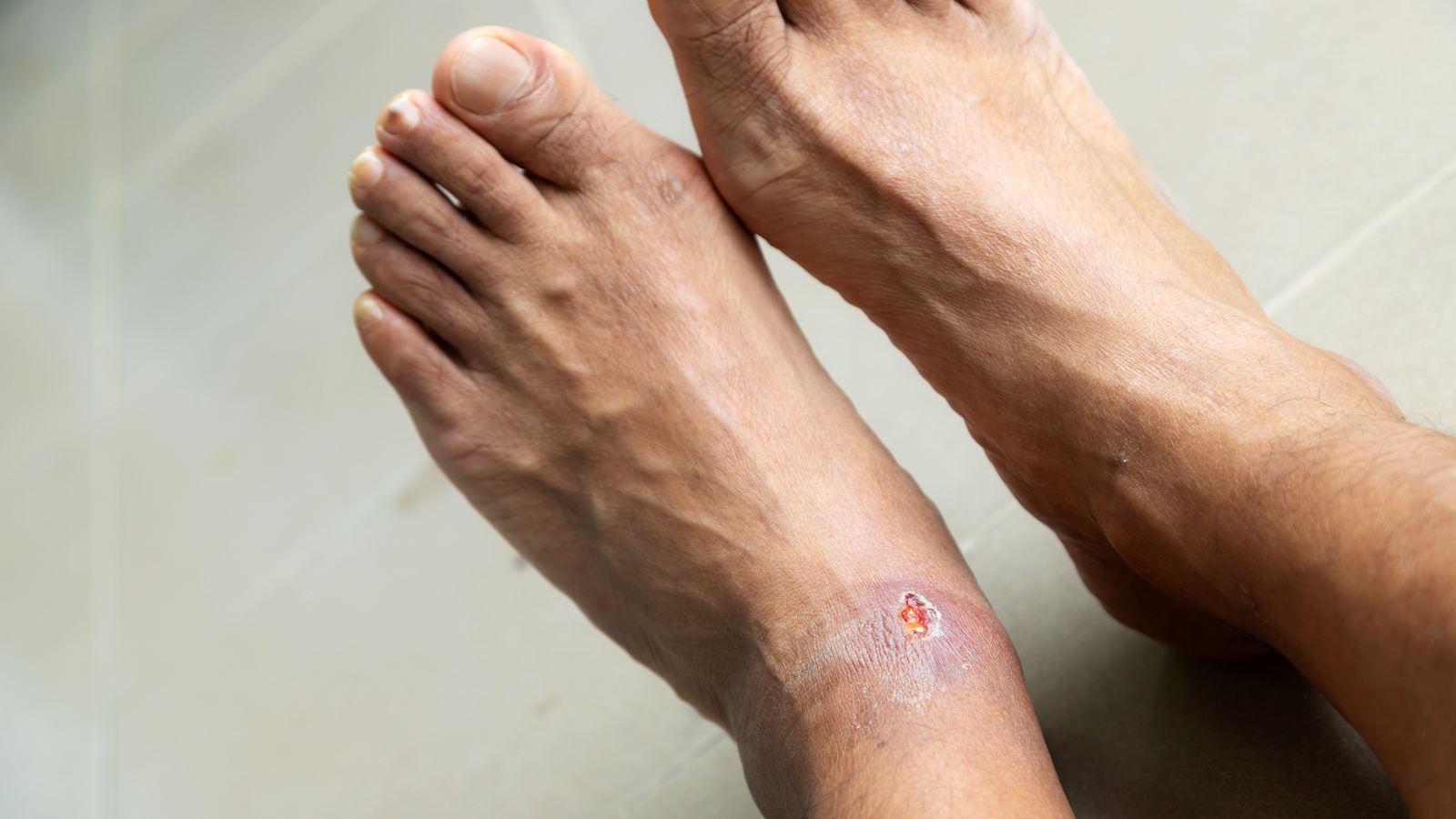
A significant breakthrough in regenerative medicine has revealed how mechanical stress at the cellular level contributes to the chronic non-healing nature of diabetic ulcers. According to a study published in Cell Death & Differentiation, researchers have mapped a regulatory axis—the Piezo1-STING pathway—that links biomechanical forces to severe metabolic dysfunction in skin cells.
The Role of Cortistatin in Mitochondrial Protection
The research demonstrates that mechanical stress in keratinocytes, the primary cell type in the skin’s outer layer, triggers the activation of the Piezo1 ion channel. This activation leads to a dangerous accumulation of glucose and advanced glycation end-products (AGEs). This metabolic overload causes mitochondrial DNA to leak into the cytosol, which subsequently activates the cGAS-STING signaling cascade. This immune-inflammatory pathway is a primary driver of tissue damage in diabetic patients.
Therapeutic Potential for Diabetic Wound Care
The study identifies the neuropeptide cortistatin (CST) as a potent inhibitor of the Piezo1 channel. By binding to Piezo1, cortistatin effectively attenuates calcium influx and prevents the downstream cascade that leads to cellular death. In laboratory models, the application of CST showed a remarkable ability to preserve mitochondrial integrity and accelerate the healing of diabetic ulcers. These findings provide a clear mechanism for how targeted therapy might one day prevent the severe complications often associated with diabetic skin damage.
- The Piezo1-STING axis acts as a bridge between physical mechanical stress and metabolic decay in diabetic skin cells.
- Cortistatin serves as an unrecognized inhibitory ligand that blocks this pathological signaling pathway.
- Targeting this regulatory axis offers a novel clinical strategy for managing chronic, non-healing diabetic wounds.
The identification of the cortistatin-Piezo1-STING axis represents a pivot from general inflammation management toward precision molecular intervention in diabetic complications, addressing the root cause of tissue degradation rather than merely managing symptoms.